False
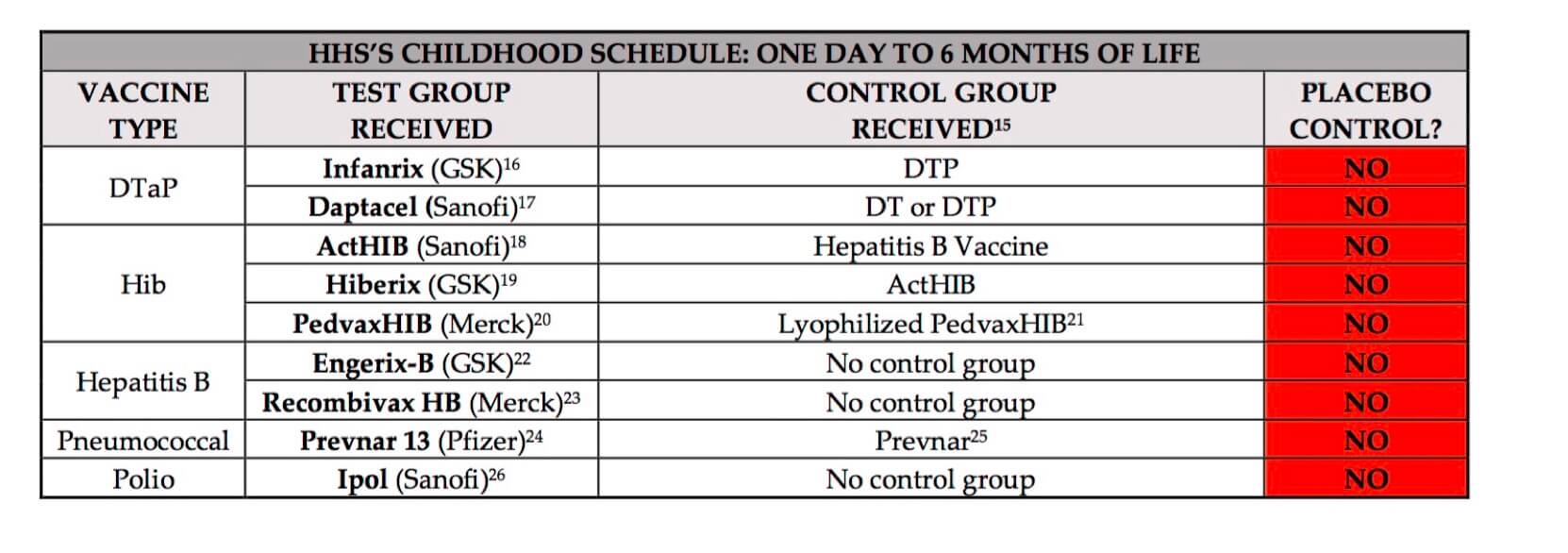
The gold standard of medical testing is the randomized controlled trial (RCT) comparing the drug to a true placebo that is completely inert. If injectable, a solution of saline (a salt water solution similar to the body’s own plasma) is usually used. Without comparing the drug to a true placebo, the testing of a new substance is incomplete.
Because vaccines are classified as biologic drugs, different rules apply than to drug testing. Hence vaccine manufacturers rarely use true placeboes in their control groups. Here are examples taken from vaccines’ ingredient inserts for US vaccines—similar to Australian vaccines. (click the image to enlarge):
- vaccines/approvedproducts/ucm124514.pdf
- vaccines/approvedproducts/ucm103037.pdf (lists DT vaccine in one of its efficacy trials as a “placebo”)
- Vaccines/ApprovedProducts/UCM109841.pdf
- Vaccines/ApprovedProducts/UCM179530.pdf
- Vaccines/ApprovedProducts/UCM253652.pdf
- In Lyophilized PedvaxHIB’s pre-licensure trials, the test group received Lyphilized PedvaxHIB, OPV and DTP, and the control group received a placebo, OPV and DTP. Ibid. Concomitantly injecting OPV and DTP negate the benefit of having a placebo as it prevents assessing the actual safety profile between Lyophilized PedvaxHIB and a placebo.
- Vaccines/ApprovedProducts/UCM224503.pdf
- Vaccines/ApprovedProducts/UCM110114.pdf
- Vaccines/ApprovedProducts/UCM574852.pdf (While a placebo was used in trials for adults over 65 years old, no placebo was used in trials to license this vaccine for children.)
- labeling.pfizer.com/showlabeling.aspx?id=134
- ApprovedProducts/UCM133479.pdf
Combinations of Vaccines
There have never been studies on the effects of the combinations of vaccines that appear on the Australian Childhood Vaccine Schedule. In addition, a comparative health outcomes study has never been conducted to determine whether the vaccine program as a whole is resulting in overall improved outcomes.
This chart reflects the massive increase in doses now given to US children. The Australian statistics are only slightly less dramatic and rapidly approaching the US numbers.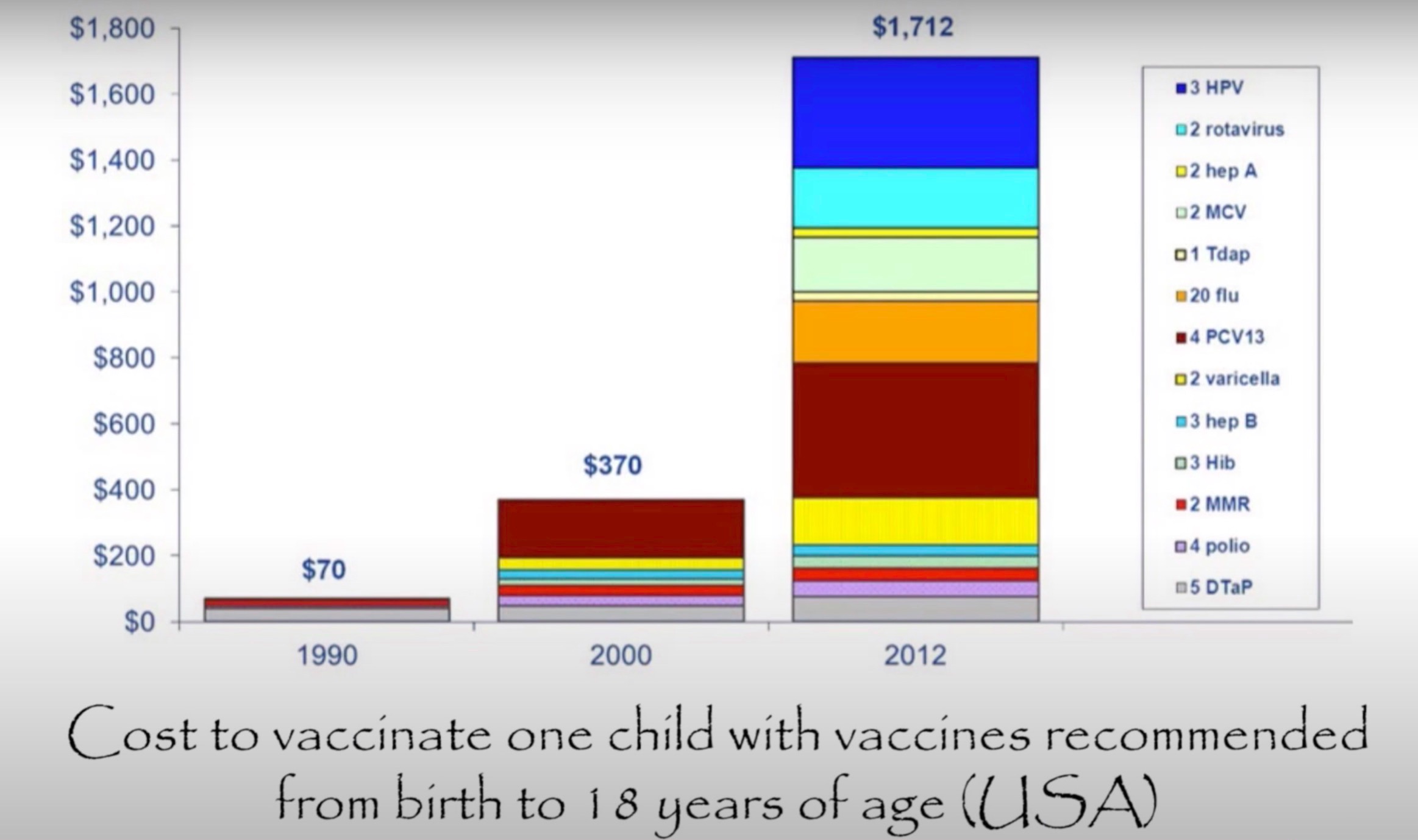
Adjuvants* and Additives
Many of the ingredients in vaccines have not been individually tested for safety by either vaccine makers or regulatory bodies. These include aluminum, polysorbate 80, and mercury (thimerosal), for which independent studies have shown toxic effects.
Vaccines are often grown in culture media of biological origin, such as the often-used human fetal tissue. The possible long-term effects of cell substrate contamination have not been thoroughly examined.
Length of Follow-up
Subjects should be followed for sufficient periods to capture long-term events, such as autoimmune diseases that can take years to develop. The pre-licensing clinical trials required to approve a vaccine are sometimes as short as a few days or weeks, making it impossible to evaluate the risk of longer-term adverse outcomes such as autoimmune illnesses.
One example is the clinical trials for Merck’s Recombivax hepatitis B vaccine, the first dose of which is administered at birth. Fewer than 150 infants and children were monitored before the vaccine was released with a follow-up that was for only five days after each dose.
* Adjuvant (from Latin, adjuvare “to help”)
A pharmacologicalor immunological agent that improves the immune response of a vaccine. Examples are aluminum, embryo cells and various chemicals.